Introduction
A post-market clinical follow-up (PMCF) report analyzed 14 cases where Neobone® was used as a bone filler in extremities for trauma management and surgical bone voids/gaps in adult patients.
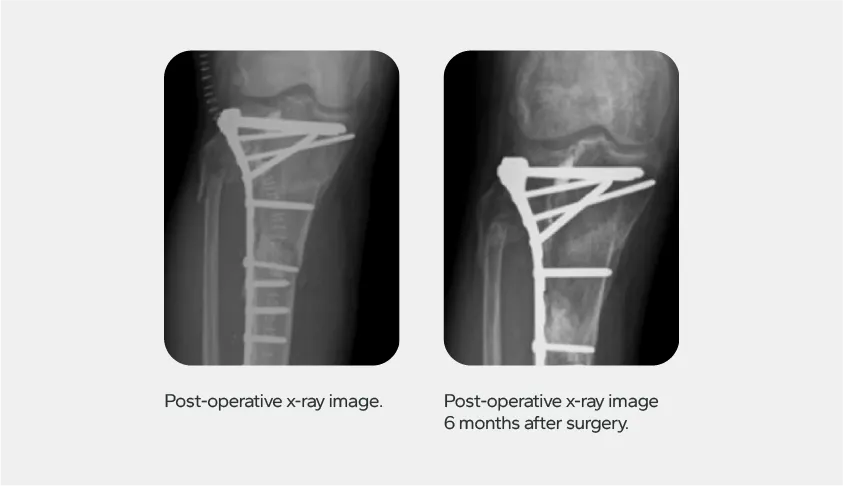
Study Overview
Diagnosis
Anatomical Distribution
Follow-up Outcomes
Safety & Complications
Conclusion
Neobone® demonstrated high safety and efficacy in orthopedic applications, showing effective bone consolidation and complete pain relief for gonalgia patients. The study found no direct device-related complications, reinforcing its reliability as a bone graft substitute in extremity surgeries.